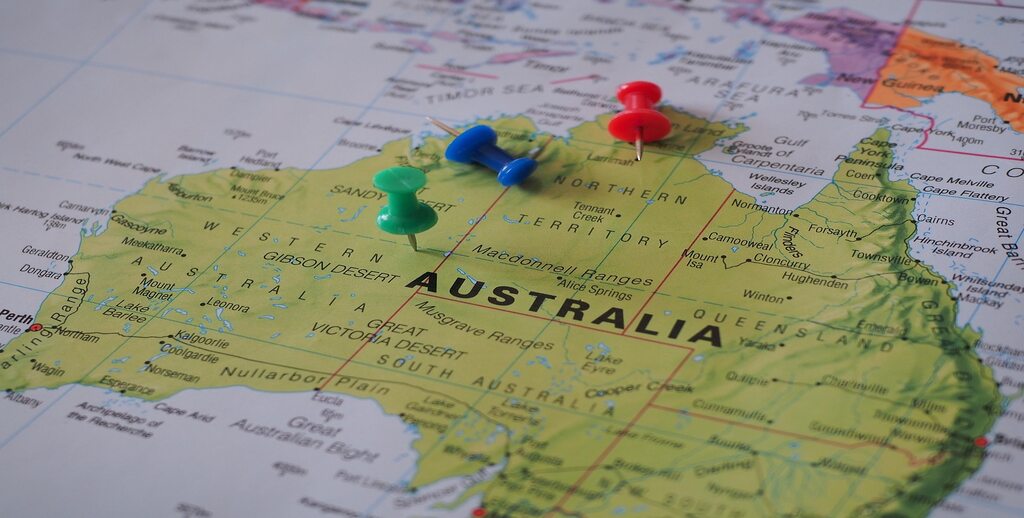
PLANT PROTECTION PRODUCTS REGULATION SERVICES IN AUSTRALIA
The regulatory authority for manufacturing, importing and supplying agrochemicals and veterinary chemicals in Australia is the Australian Pesticides and Veterinary Medicines Authority (APVMA). The Agricultural and Veterinary Chemicals Act, 1994 is an Australian federal law that governs the regulation of agricultural and veterinary chemicals. The APVMA regulates different categories which include, but are not limited to Pesticides, Biopesticides, Household pesticides, Veterinary chemicals and products.
Application Types
The application types include, but are not limited to, the following types:
- Active constituent approvals
- Product registration
- Additional labels for a registered product
- Variations to registered actives or products
- Timeshift applications
- Renewal of registration
- Permit application
Key Services:
Strategic advice and guidance: Auxilife's regulatory expertise helps you understand the registration status of similar agrochemical active ingredients and products in Australia, relative to applicants intended product. We'll guide you in determining the correct application item number and data requirements for your agrochemical registration.
Support in nominating an agent (legal entity): For overseas applicants, appointing a nominated agent is a mandatory requirement with the APVMA. Auxilife offers the service of establishing this legal entity in Australia, fulfilling the nominated agent role.
APVMA Online Service Portal: Auxilife offers a dedicated service to assist you in the seamless creation and setup of your account on this essential platform, ensuring you can efficiently manage your regulatory submissions.
Data Gap Analysis: Our team meticulously analyses all relevant information to ensure a seamless registration process. We expertly identify and address critical gaps in your data across key areas, including Chemistry, Health, Residue and Trade, and Environment, Efficacy, and Safety. This comprehensive approach optimizes your submission for successful approval.
Data generation management & Study monitoring: Auxilife provides expert consultancy and support for data generation, study monitoring, and study plans in collaboration with GLP labs worldwide.
Placing and monitoring efficacy/residue trials: Our team specializes in designing efficacy field trials that meet APVMA's requirements, ensuring comprehensive and compliant data.
Application preparation including dossier preparation: Crafting a compliant dossier, encompassing all administrative and technical modules, is a complex undertaking. Auxilife streamlines this intricate process by offering expert regulatory services that include comprehensive dossier preparation and accurate application form submission, all meticulously aligned with the Agvet Code and relevant regulations.