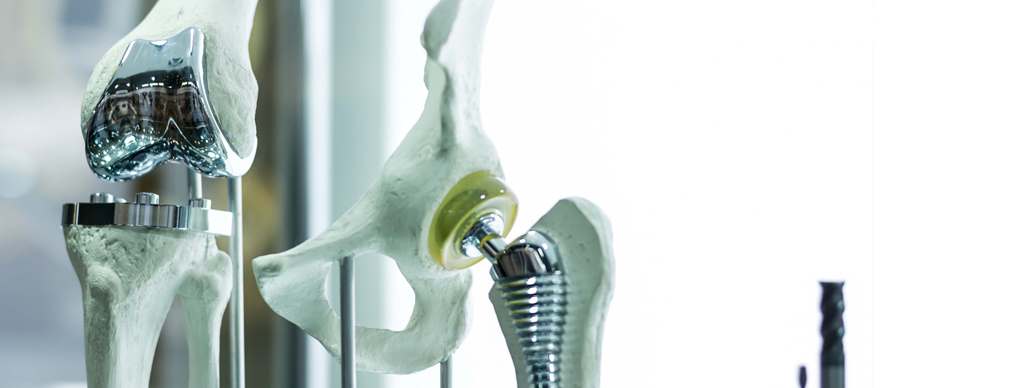
MEDICAL DEVICES REGULATION SERVICES IN INDIA
Central Drug Standard Control Organization (CDSCO) has released the Medical Devices amendment rule which is effective from 1st April 2020.
CDSCO has released classification of non-notified medical device and In-vitro diagnostics (IVD’s) on 3 September 2020, devices have been classified into 24 categories of medical devices and 3 Categories of In-Vitro Diagnostics.
The registration is on voluntary basis for a period of eighteen months (1st April 2020 to 30th Sep 2021), from the commencement of this rule, after which (from 01 October 2021), it shall be compulsory.
After the end of voluntary registration period, it shall be mandatory to obtain the registration number and it would be done in phase wise manner. Auxilife offers a skilled guidance for acquiring import and manufacturing licenses for the sale of medical devices in India.
However, separate licenses are required to be obtained for the manufacture and import of medical devices as separate conditions are enforced to be fulfilled for obtaining these licenses. There are various classes of devices such as:
- Class A- low-risk
- Class B- moderate, low-risk
- Class C- moderate, high-risk
- Class D- high-risk
Key Services
- Identify the category according to the new classifications of the medical devices based on the intended purpose.
- Guidance for the process to obtain the import license/manufacturing licence.
- Guidance for conformance with Quality Management System.
- Guidance for obtaining permission to conduct clinical trials as required.
- Compile the application and all necessary documents for the online registration.
- Coordinate the medical device registration approval with the CDSCO.
FAQ
- How to register medical devices in India?
- Medical device registration in India?
- Indian medical device regulatory compliance?
- What is the registration process of medical device in India?
We support you to get answer for all these questions so you can prepare a robust strategy. Let’s connect with us today!
